How much actual root is in “root vegetables”?
The wintertime pantry is a study in vegetable dormancy. Our shelves brim with structures plants use to store their own provisions. Each embryonic plant in a seed—the next generation of oats, quinoa, dry beans, walnuts—rests in the concentrated nutritive tissue gifted to it by its parent. The starchy flesh within the impervious shell of a winter squash is alive, as are apples, hopeful vessels of seed dispersal. Maple and birch syrup are stored energy made liquid and bottled. And then there are the so-called “root vegetables.” The term covers a surprisingly anatomically varied set of nutrient storage structures, only some of which are actual roots. Our familiar root vegetables represent only a sliver of global plant species diversity but nonetheless include the majority of contrivances herbaceous plants use in order to live to sprout another season: taproots, hypocotyls, stem tubers, root tubers, corms, and rhizomes. Raiding your root cellar for the ingredients for a roasted root vegetable medley, then, provides a great opportunity to turn your dinner prep into a botany lab. All you need is a knife and cutting board.
The case for tree thinking
First we need to consider the taxonomy of our candidate botanical subjects. Taxonomy is the scientific practice of grouping related organisms in hierarchies of similarity. We shove the continuous variation of living things into discrete boxes labeled species, genus, family, order, and so on. Carl Linnaeus started the taxonomic naming system two hundred years before Watson and Crick identified the double helix shape of deoxyribonucleic acid (DNA), marking the beginning of the genomics era. Modern practitioners bring many types of data to bear—geography, fossils, genetics, morphology—toward the twin goals of illuminating the pattern of plant species evolution and defining groups based on common ancestry.
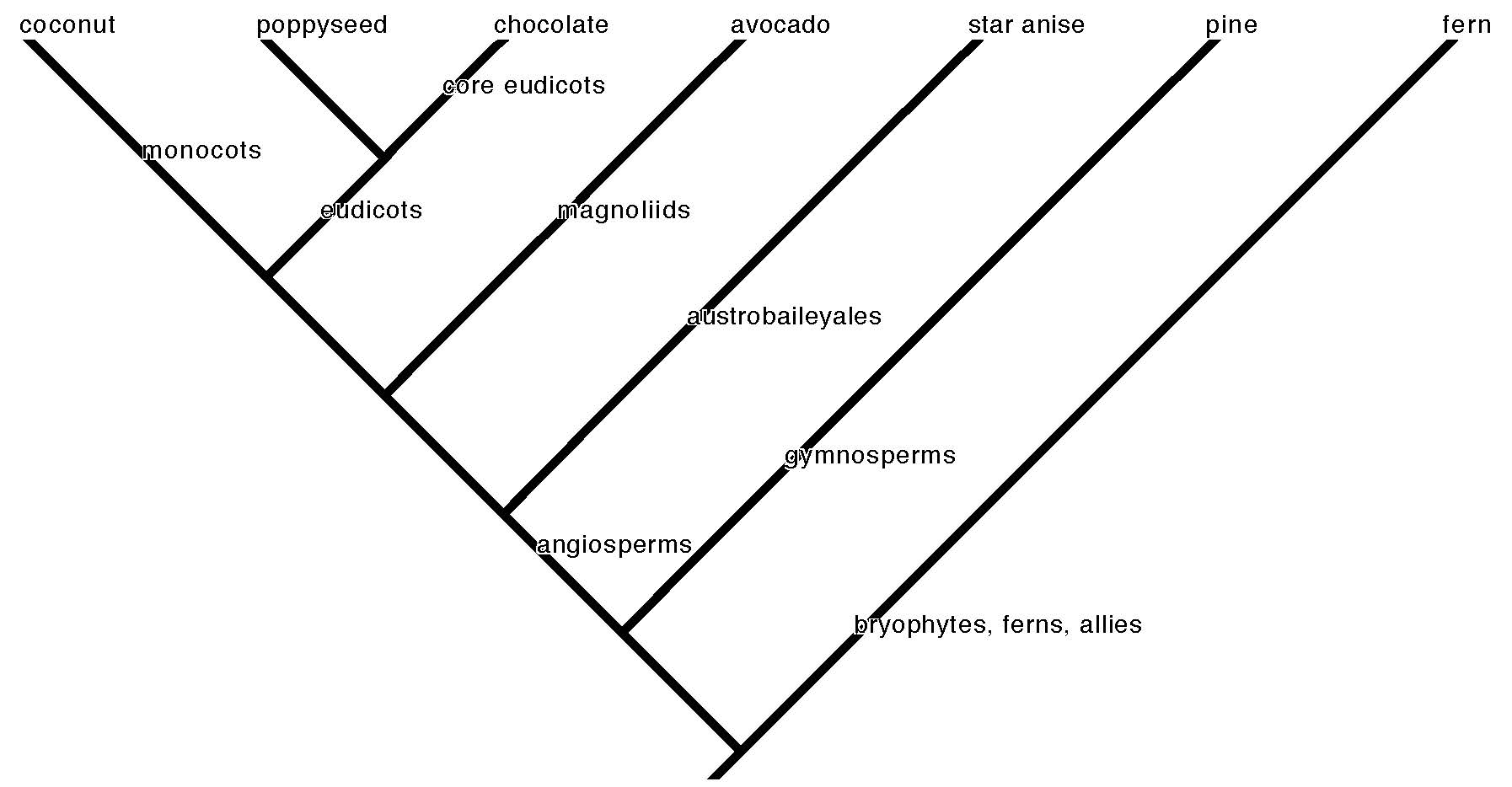
The visual embodiment of this effort is the tree of life (cladogram) that represents the pattern of plant species evolution by common descent (phylogeny–see our primer on reading phylogenetic trees and using them to understand broad patterns in plant evolution). Each branching point on the tree is a node that represents a common ancestor of all the descendant taxa on the branches that come from it. The species are like the leaves on the tips of the branches. A schematic tree of life is the only illustration in Charles Darwin’s On the Origin of Species, the landmark book that provided the kernels of the core theories of evolutionary biology. Modern scientific convention tries to match old taxonomic names—because they are familiar and useful as a practical matter—with nodes on the tree of life. Small branches connect species to genus. Larger branches connect genera to families, families to order. The deep internal named nodes show the origin of the major clades. A clade is a group of organisms that descend from a common ancestor. A major clade is a significant branch on the plant tree of life that scientists have named for convenience of reference. You may remember some of these from biology class, like “monocot” and “dicot.” The former (monocots) has held up as a robust clade, but dicot is more complicated.
As it happens, plant taxonomy before the advent of genetic data was reasonably accurate. Even though our understanding of plant species evolution is far from complete, genomic analysis has provided few big surprises about common ancestry of plant species and membership of taxonomic groups. Early taxonomists had the wisdom to rely primarily on similarity of reproductive structures—seeds, fruit, flowers, spores, cones—to circumscribe named groups. Reproductive structures tend to change more slowly over evolutionary time than do vegetative structures in plants. So one may expect to find a fair amount of coincident similarity among distantly related species in roots, shoots, and leaves.
This is where our categorization of root vegetables by taxonomy collides with our categorization of them by morphology. In short order we will organize our root vegetable species according to which structures the plant has chosen to amplify as a subterranean or soil-adjacent storage organ. This is not the same pattern as taxonomic organization. Grouping our root vegetables by taxonomy first helps us understand similarity and difference within and between groups of closely related plants—families, in this case. In doing so we can develop gestalt for the culinary qualities within plant families and appreciation for the evolution of plant diversity evident on our own dinner tables. Consider this intellectual nourishment, or perhaps the advent of a lens with which to view familiar foods anew.
Placing root vegetables on the plant tree of life
Around the globe humans utilize many dozens of plant species that bear underground (or near enough) storage structures. The most recent generations of people overwintering in the United States or Europe, however, chiefly engage with only a few. Perhaps only the most dedicated winter vegetable enthusiast will be familiar with all of the species on the following roster of root vegetables potentially available in Western grocery stores or farmer’s markets, although the list is unlikely to be exhaustive. I have organized the root vegetable species by families, and the families by major clade. Our list includes 15 of the 446 currently recognized plant families.

Please take note of the disambiguation about the words “yam” and “potato.” The tubers marketed as “yams” in most American groceries are mostly actually sweet potatoes, which are also not potatoes. True yams are large tubers that are staples of tropical diets but relatively scarce in northern diets or groceries. In New Zealand the Andean oca is also known as “yam.” All of these are in different plant families.
Monocots:
- Amaryllis family (Amaryllidaceae): onions and shallots (Allium cepa), garlic (Allium sativum), leeks (Allium ampeloprasum), and other alliums
- Ginger family (Zingiberaceae): ginger (Zingiber officinale), turmeric (Cucurma longa)
- Dioscoreaceae: true yams (several species in the genus Dioscorea), including the purple yam (ube; D. alata).
- Sedge family (Cyperaceae): water chestnut (Eleocharis dulcis)
- Arum family (Araceae): taro (Colocasia esculenta)
Eudicots: asterids
- Goosefoot family (Amaranthaceae): beets (Beta vulgaris)
- Sunflower family (Asteraceae): salsify (Tragopogon porrifolius), burdock root (Arctium lappa), sunchokes (Helianthus tuberosus)
- Carrot family (Apiaceae): carrots (Daucus carrota), parsnips (Pastinaca sativa), parsley root (Petroselinum crispum), celery root (Apium graveolens)
- Morning glory family (Convolvulaceae): sweet potatoes (Ipomoea batatas; often mistakenly called “yams” in the United States)
- Nightshade family (Solanaceae): potatoes (Solanum tuberosum)
Eudicots: rosids
- Spurge family (Euphorbiaceae): cassava (manioc, yucca; Manihot esculenta), the source of tapioca
- Mustard family (Brassicaceae): turnips (Brassica rapa), rutabagas (Brassica napus), kohlrabi (Brassica oleracea), radishes (genus Raphanus), horseradish (Amoracia rusticana), wasabi (Eutrema japonicum), maca (Lepidium meyenii)
- Nasturtium family (Tropaeolaceae): mashua (Tropaeolum tuberosum)
- Legume family (Fabaceae): jicama (Pachyrhizus erosus)
- Oxalis family (Oxalidaceae): oca (Oxalis tuberosa), an Andean vegetable that is confusingly called “yam” in New Zealand
True taproots: carrot, parsnip, parsley root, salsify, burdock root, horseradish

Now grab a carrot, parsnip, parsley root, salsify, or burdock root for your roast vegetable medley. These are the only true taproots on our list. The roots are much longer than they are wide and taper to a point. Thin lateral roots sprout from them in random locations or in discrete vertical lines. If you cut it open and examine it in cross section you see the tough core xylem (water conducting tissue) in the middle surrounded by a cortical layer (cambium) that separates the core from the sweet storage tissue (parenchyma) and sugar-moving phloem that surrounds it. Structurally supportive ray fibers radiate like spokes from the core. You have likely removed the aboveground greenery from these plants but should be able to tell or recall that it appears as if the leaves grow directly out of the crown of the taproots. They almost do. The anatomical stem on carrots and parsnips is a highly reduced disk on top of the taproot that serves as a bud-studded vascular transfer station, shuttling water and nutrients from the taproot into the leaves and flowering shoots.
Horseradish is also a taproot. A little bit grated into a sauce would make a delicious accompaniment to your roast vegetable medley. Incidentally, horseradish powder is the main ingredient in cheaper “wasabi” products available in American grocery stores, as the horseradish taste is superficially similar to that of true wasabi, which is also in the mustard family. Wasabi is also a root vegetable, but its underground storage structure is a rhizome, an underground stem, not a taproot.
Roots fused with stems (hypocotyls): celery root, beet, rutabaga, turnip, radish
A hypocotyl is a swollen fusion of taproot and stem base. The taproot portion is covered in fibrous secondary roots, most spectacularly in celery root. Leaf scars will be visible about these lateral roots, either surrounding the entire upper portions of the hypocotyl, as in celery root, or just at the top, as in beets and the mustard family hypocotyl vegetables (turnip, rutabaga, radish). All hypocotyl vegetables aside from beets are structurally straightforward but different from the taproots. A single layer of vascular tissue lays below the skin surface and penetrates into the storage tissue.
Beets, however, are built from concentric rings of vascular tissue (xylem and phloem) and storage tissue (parenchyma), which is visible when the beet is cut in cross section. This ring structure is unique to the taxonomic order Caryopyllales, of which beets are a member. And as Katherine notes in her excellent beet post, the vibrant colors and earthy smell of beets are also unique. The former is due to betalain pigments, which are also unique to the Caryophyllales and distinct from the anthocyanin pigments present in all the other vegetables in our list (see our pigments post for a quick rundown of the most common pigments). The earthy smell is from a compound called geosmin. Beet is the only plant known to make it, and nobody knows why. Geosmin us usually produced by microbes in the soil and is liberated after rain to create that marvelous fresh smell after a storm.
Indidentally, our hypocotyl root vegetables here are all varieties, or subspecies, of species that also produce familiar leafy vegetables: rutabagas and the Russian or Siberian kales; turnips and Napa cabbages and mizuna; beets and Swiss chard; celery root and celery stalks or seeds. In each of these cases the variety produced for leaves has a much less pronounced hypocotyl than the variety produced as a root vegetable. Similarly, while the leaves on our hypocotyl root vegetables are all edible, they will be smaller and tougher than those on the varieties that have been bred for leaves.
Bulbs: onion, garlic, shallots, leek
Onions, shallots, garlic and other alliums might be the most famous “root vegetables” of all, but their delicious parts are constructed entirely of swollen modified leaves. The papery tunicate covering surrounding the fleshy leaf bases are also constructed out of modified leaves, all arising from the basal plate (true compressed stem) that interfaces with the spindly roots on the bottom. The fleshy part of each garlic clove is a single fat modified leaf. Inside each garlic clove or onion bulb is an apical bud that will send up new leaves and flowering shoots. Everyone who has had onions and garlic sprout on them can observe this. You can of course plant these sprouting bulbs in the soil to make a new plant. A leek is a bit intermediate between a true bulb and a big herb. They call the lower white region of overlapping succulent leaf bases a “pseudobulb,” a nod to the messy continuous nature of biology and the difficulty with labels.
Unless you’re using a variety of “sweet” onion, which has been grown or bred to lack sulfurous aromatic compounds, you might tear up when you’re cutting onions and shallots. Cutting these bulbs volatilizes the irritating compounds that otherwise protect our favorite bulbs from pests.
Root tubers: sweet potatoes, cassava
A root tuber is an enlarged root that stores starch and other nutrients. Smaller lateral roots often branch from its surface and obtain water and soil nutrients. Raw sweet potatoes are readily available candidate root tuber ingredients for your botanical scrutiny and roast vegetable medley. Cassava is not, nor should it be, at least in root tuber form. Starch derived from cassava might be elsewhere in your pantry as tapioca.
A convenient aspect of our most commonly used root vegetables is that they require very little manipulation or preparation before they can be consumed. You don’t even have to peel your sweet potatoes before you cook them. Raw cassava tubers, however, are laced full to bursting with cyanide. They are the third most important source of calories throughout the tropics, behind corn and rice, but require extensive preparation before consumption to remove the cyanide, including grating, drying, leaching and cooking.
Cassava tubers develop underground from certain roots that become fleshy storage structures. They continue to acquire water and nutrients via smaller secondary roots that dot their surface. If the plant in question grows from a seed, then the harvestable storage root may develop from the taproot that grows from the seed. This, however, proves an inefficient way to farm these species, as many more storage roots can develop on a single plant when that plant is started from a shoot—a stem with leaves. This is where the visual heuristic of placing root vegetable species on the branches of the plant tree of life gets literal with sweet potatoes and cassava. The key factor is the presence of numerous nodes—leaves along the stem and their attendant axillary buds. Cassava and sweet potato are among the plant species that can generate roots from the buds in their leaf axils under the right conditions, namely being in contact with moist soil. Roots that develop from non-root tissue (like stems) are called adventitious roots. When several nodes of a shoot are planted in the soil, many adventitious roots will develop, of which some can become enlarged storage roots. In cassava the starting shoot is a cutting from a mature cassava plant. In sweet potatoes the starting shoot is called a slip. Slips grow from buds on the proximal (closest to the parent plant) end of sweet potato tubers. On sweet potatoes this is the end with the scar where the tuber was cut away from the parent plant.
Rhizomes: turmeric, ginger, galangal, lotus, arrowroot, wasabi
A rhizome is a fleshy underground stem. It grows horizontally and sprouts new plants. Stems grow upward from buds near the soil surface, and roots grow from buds on the underside of the rhizome. It is structurally similar to stem tubers, like potatoes (see below), but it only grows horizontally, not in any direction, like a tuber. Rhubarb, asparagus, and irises also spread by rhizomes. If you decide to get out ginger or turmeric to flavor your vegetable medley, you’ll notice structural similarities to stem tubers, including nodes with buds.
Stem tubers: potato, sunchoke, jicama, yam
The eyes may or may not be the window into the soul, but they are our most conspicuous clue that potatoes are subterranean stem tubers, not roots. Katherine’s superb post on potato anatomy will walk you through this (potato) eye exam. Observe both ends of a potato. One end (the proximal end) bears the stump of the stolon (horizontal stem) that connected it to its mother plant. The other is tightly packed with small eyes that spiral out and around the potato. This is the growing (distal) end of the potato. New eyes originate at this end, so each eye is progressively older as you move toward the middle of the potato. Each eye contains a cluster of buds subtended by a semicircular leaf scar. The leaf in question was vestigial, translucent, and a remnant of it may still be present on your potato. Eyes are most easily visible on the “waxy” potato varieties (like Yukon Golds), which have less starch overall and a different ratio of types of starch than the “starchy” varieties (like Russets)–see Katherine’s post on potato starchiness for details.
The buds in each eye are axillary buds, structurally the same as Brussels sprouts. If your potato is exposed to enough light or warmth, the axillary buds will grow into new leafy stems, each of which can create a new potato plant. In this case your potato might also start synthesizing chlorophyll, turning it green. It will make toxic compounds at the same time, though, so if your potato is green you should either liberally peel it or wait to plant it in the spring.

Nodes and buds are also easily visible on sunchokes, less so on jicama. True yams are actually structurally intermediate between rhizomes and stem tubers in that they might sprout adventitious roots. If you get your hands on an actual yam, instead of a sweet potato, you might see these.
Corms: taro, water chestnut (with a note on kohlrabi, which is not a corm)
A corm is yet another method by which plants have modified their stems to store starches and nutrients underground. The storage tissue is a swollen area of the stem above the roots and below the apical bud, from which leaves and flowers develop. Lateral buds on the stem produce modified leaves that produce a protective tunicate sheath around the starchy corm tissue. A thickened basal plate on the bottom interfaces with the roots and may sprout new corms (cormels). If you get canned water chestnuts or taro corms for your vegetable medley, these structures should be visible to you. Structurally, a taro corm is most similar to kohlrabi, which is what happened when plant breeders long ago took a weedy ancestral cabbage plant (Brassica oleracea) and bred for fat, bulbous stems. The leaf scars out the outside of a kohlrabi, and the nubbin of a root on the bottom, reveals that it is entirely stem.
The geophyte lifestyle
Potatoes are in the same genus (Solanum) as tomatoes (S. lycopersicum) and eggplants (S. melongena). The potato is the only one of these close relatives that hails from high in the Andes, where its underground tubers store the starches it needs to survive the harsh alpine conditions. This is a common ecological theme. Plants that create underground storage organs to withstand winter or seasons of drought are called geophytes. Even our short list of root vegetable species demonstrates that the geophyte lifestyle independently pops up all over the plant evolutionary tree, presumably in times and places where it may be adaptive. Even in just the Andes alone, potatoes are not the only domesticated geophyte crop with lowland relatives in the same genus devoid of starchy storage organs. Oca, confusingly called “yam” in New Zealand, where it was introduced in the mid-19th century, is otherwise known as Oxalis tuberosa. It makes stem tubers, like a potato. The specific epithet “tuberosa” separates it from non-geophyte species of Oxalis that are probably familiar to hikers and gardeners throughout the northern hemisphere. American health food stores sell dried maca hypocotyl (Lepidium meyenii) as a health food supplement, even though it is a staple crop throughout montane South America. Other Lepidium species are weedy little mustard plants. In the summer your garden may be teeming with flowering nasturtiums (Tropaeolum majus). You’ll notice a distinct lack of a fat, starchy stem tuber. Not so with mashua (Tropaeolum tuberosum).
You should be well on your way at this point to getting your root vegetable medley into the oven. Finish peeling your vegetables, if you must, and dice them into approximately equally sized chunks. Toss them with a small amount of oil and salt. Add herbs if you would like. Spread them in a single layer on a baking tray or roasting pan and roast in the oven at 375 degrees Fahrenheit until they are tender, about 40 minutes. It is helpful to turn the pieces and move them around with a metal spatula halfway through the cooking time.
I like to serve these roast vegetables with some kind of sauce, often a strained yogurt mixed with salt and herbs. This is a dish filled by design with concentrated energy to maintain life through harsh seasons. The geophyte lifestyle is periodically useful for us all.